News und Analysen
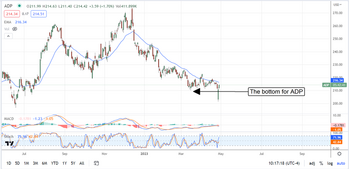
Dividend Prince Automatic Data Processing Hits Bottom
If you’ve been wondering when Automatic Data Processing (NASDAQ: ADP) will hit bottom, it looks like it is in. The pre-Q3 earnings release price action and the report scream bottom and have the

Fiserv's Growth Prospects Shine As Other Financials Slump
Whenever an entire sector appears to be on fire, as is the case with financials at the moment, there are usually areas of opportunity among stocks bucking the broad sector trend.
One stock

Microsoft Crushes; Get Excited
A 7% jump in last night's after-hours session should tell you everything you need to know about Microsoft (NASDAQ: MSFT) Q2 earnings. We'll have to wait and see how shares trade in Wednesday's

Here's Why Google's Report Is Just Good Enough
Last night, Alphabet Inc (NASDAQ: GOOGL), the parent company of Google, announced its first-quarter earnings, revealing strong financial results that beat both market and analyst expectations

PepsiCo Bubbles To All-Time High, More to Come
If The Coca-Cola Company (NYSE: KO) and PepsiCo (NASDAQ: PEP) were just about beverages, then The Coca-Cola Company would be the better choice. It is the larger of the 2 regarding its position in

Why Legend Biotech Stock Is Having Its Best Month Yet
Legend Biotech Corporation (NASDAQ: LEGN) is among the many healthcare names vying for a breakthrough in cancer treatment. Last week, the New Jersey-based company had just that.
On April 19th, a
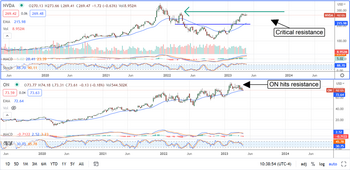
Is it Time to Take Profits in Chip Stocks?
Most chip stocks are up off their lows and have trended higher, like NVIDIA Corporation (NASDAQ: NVDA) and On Semiconductor Corporation (NASDAQ: ON). The first quarter season should be solid amid

2 Industrial Strength Dividend Stocks Melting Up
Industrial stocks have been struggling to gain traction in 2023, but news from PPG Industries (NYSE: PPG) and CSX (NYSE: CSX) has these stocks melting-up. The combination of underlying business

Moderna: Europäische Arzneimittelagentur erteilt für MRNA-4157/V940, einen personalisierten MRNA-Prüfimpfstoff gegen Krebs, in Kombination mit Keytruda(R) (Pembrolizumab) den Status eines prioritären Arzneimittels (PRIME) für die adjuvante Behandlung
Europäische Arzneimittelagentur erteilt für MRNA-4157/V940, einen personalisierten MRNA-Prüfimpfstoff gegen Krebs, in Kombination mit Keytruda(R) (Pembrolizumab) den Status eines prioritären

Moderna: MRNA-4157/V940, an Investigational Personalized MRNA Cancer Vaccine, in Combination With Keytruda(R) (Pembrolizumab), Receives Prime Scheme Designation From the European Medicines Agency for Adjuvant Treatment of Patients With High-Risk Stag
MRNA-4157/V940, an Investigational Personalized MRNA Cancer Vaccine, in Combination With Keytruda(R) (Pembrolizumab), Receives Prime Scheme Designation From the European Medicines Agency for

Moderna finalisiert Abkommen mit Regierung von Republik Kenia hinsichtlich Errichtung von mRNA-Fertigungsanlage
- Anlage soll Kenia und afrikanischem Kontinent Zugang zu mRNA-Impfstoffen verschaffen, Gesundheitssicherheit gewährleisten und auf globalem Engagement von Moderna in Bereich von

Moderna Finalizes Agreement with the Government of the Republic of Kenya to Establish an mRNA Manufacturing Facility
- Facility to enable access to manufactured mRNA vaccines for Kenya and the African continent, providing health security and building upon Moderna's global public health commitments

Chantal Friebertshäuser zu Senior Vice President, Commercial von Moderna ernannt
Neu geschaffene Position wird für kommerzielle Bestrebungen in Europa, Nahen Osten und Kanada verantwortlich sein
Cambridge (Massachusetts) / Accesswire / 20. Dezember 2022 / Moderna Inc.

Chantal Friebertshäuser Joins Moderna as Senior Vice President, Commercial
Newly created role will have responsibility for commercial efforts in Europe, Middle East and Canada
CAMBRIDGE, MA / ACCESSWIRE / December 20, 2022 / Moderna, Inc. (NASDAQ:MRNA), a

EMA Committee for Medicinal Products for Human Use (CHMP) Recommends the Use of Moderna’s BA.1 Targeting Bivalent COVID-19 Booster in Children (6-11 Years) In the European Union
The recommendation is based on clinical data for Moderna's bivalent Omicron-targeting COVID-19 vaccine, mRNA.1273.214
Moderna's bivalent Omicron-targeting COVID-19 vaccines (mRNA.1273.214

Der Ausschuss für Humanarzneimittel (CHMP) der EMA empfiehlt die Anwendung von BA.1 Targeting Bivalent-COVID-19-Auffrischimpfung von Moderna bei Kindern (6–11 Jahre) in der Europäischen Union
Die Empfehlung stützt sich auf klinische Daten zum bivalenten auf Omikron zugeschnittenen COVID-19-Impfstoff von Moderna, mRNA.1273.214
Die an die Omikron-Virusvarianten angepassten

Moderna and Merck Announce mRNA-4157/V940, an Investigational Personalized mRNA Cancer Vaccine, in Combination With KEYTRUDA(R) (pembrolizumab), Met Primary Efficacy Endpoint in Phase 2b KEYNOTE-942 Trial
mRNA-4157/V940, in combination with KEYTRUDA, demonstrated a statistically significant and clinically meaningful reduction in the risk of disease recurrence or death compared to KEYTRUDA

Moderna und Merck geben bekannt, dass mRNA-4157/V940, ein personalisierter mRNA-Krebsimpfstoff, in Kombination mit KEYTRUDA(R) (Pembrolizumab) primären Endpunkt in Phase-2b-Studie KEYNOTE-942 erreicht hat
mRNA-4157/V940 zeigte in Kombination mit KEYTRUDA statistisch signifikante und klinisch bedeutsame Verringerung von Risiko für Wiederauftreten der Erkrankung oder Tod im Vergleich zu

Moderna Inc.: EMA Committee For Medicinal Products For Human Use Adopts Positive Opinion Recommending Authorization Of Moderna's Omicron BA.4-BA.5 Targeting Bivalent Covid-19 Vaccine In The European Union
Positive recommendation follows the recent approval of Spikevax bivalent Original/Omicron BA.1 (mRNA-1273.214), a bivalent booster vaccine targeting the Omicron BA.1 subvariant, in

Moderna Inc.: Der EMA-Ausschuss für Humanarzneimittel nimmt positive Stellungnahme an, in der die Zulassung des bivalenten, auf Omikron BA.4-BA.5 abzielenden Covid-19-Impfstoffs von Moderna empfohlen wird
Die positive Empfehlung folgt auf die kürzlich erfolgte Zulassung von Spikevax bivalent Original/Omicron BA.1 (mRNA-1273.214), einem bivalenten Booster-Impfstoff, der sich gegen die

Moderna Inc.: Der EMA-Ausschuss für Humanarzneimittel gibt eine positive Stellungnahme, in der die Zulassung zur Verwendung von Spikevax (mRNA-1273) bei Kindern von 6 Monaten bis 5 Jahren in der Europäischen Union empfohlen wird
Die Ankündigung folgt auf die vorherige Entscheidung des CHMP, eine positive Stellungnahme abzugeben, in der die Zulassung für den COVID-19-Impfstoff von Moderna für Kinder ab 6 Jahren

Moderna Inc.: EMA Committee for Medicinal Products for Human Use Issues Positive Opinion Recommending Authorization for the Use of Spikevax (mRNA-1273) in Children 6 Months - 5 Years in the European Union
Announcement follows CHMP's previous decision to issue a positive opinion recommending marketing authorization for Moderna's COVID-19 vaccine to include children 6 years of age and older

Merck und Moderna geben bekannt, dass Merck seine Option für die gemeinsame Entwicklung und Vermarktung eines personalisierten Krebsimpfstoffs im Erprobungsstadium ausübt
Die Unternehmen werden planmäßig im 4. Quartal 2022 Datenmaterial aus der laufenden Phase-II-Studie zu mRNA-4157/V940 in Kombination mit KEYTRUDA® als adjuvante Therapie beim

Merck and Moderna Announce Exercise of Option by Merck for Joint Development and Commercialization of Investigational Personalized Cancer Vaccine
Companies on track to report data from the ongoing Phase 2 trial of mRNA-4157/V940 in combination with KEYTRUDA® as adjuvant therapy in high-risk melanoma in 4Q 2022
CAMBRIDGE, MA and

Netflix, Inc. geht in Seitwärtstendenz über – Chancen für Inliner
Nachdem die Netflix-Aktie am Hoch des Jahres 2022 scheiterte, ist eine Korrektur erfolgt, die bis heute andauert. Unterstützung und Widerstand funktionieren laut Charttechnik. In diesem Fall dürfte




