News

EQS-News: Newron to present at the 31st European Congress of Psychiatry

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc
EQS-News: PAION IS HOSTING A SPONSORED ANGIOTENSIN II SYMPOSIUM AT ISICEM IN BRUSSELS

EQS-News: Evotec announces progress in strategic protein degradation partnership with Bristol Myers Squibb
EQS-News: MorphoSys AG Reports Fourth Quarter and Full Year 2022 Financial Results and Provides Corporate Update

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc

EQS-Adhoc: GN Store Nord A/S: Update on the agenda for GN Store Nord’s Annual General Meeting and capital raise
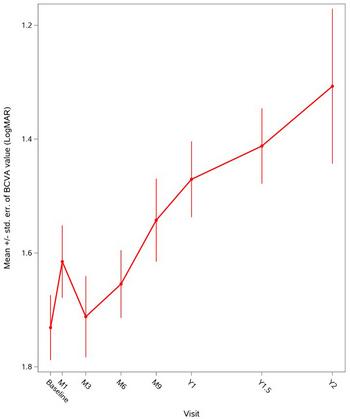
GenSight Biologics Announces Presentation of LUMEVOQ® Efficacy and Safety Data from Early Access Programs for ND4-LHON Patients at NANOS 2023
Regulatory News:
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20230314006021/en/
Figure 1: Global evolution of mean BCVA over two
EQS-News: Lucinda Crabtree to Join MorphoSys as Chief Financial Officer

Geisinger and Acadia Healthcare Appoint Kelly Ankenbrand as CEO of Geisinger Behavioral Health Center Northeast
Geisinger and Acadia Healthcare have named Kelly Ankenbrand, M.B.A., as chief executive officer for Geisinger Behavioral Health Center Northeast, a new 96-bed inpatient behavioral health hospital

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc
EQS-News: Biotest AG: Biotest donates human albumin for earthquake victims in Turkey

EQS-News: Newron announces 2022 financial results and provides outlook for 2023

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc
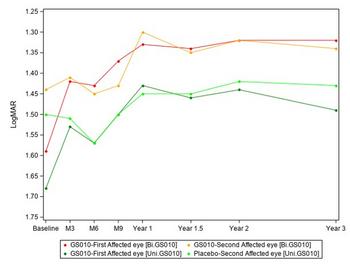
GenSight Biologics Confirms Sustained Efficacy and Safety of Bilateral LUMEVOQ® Injections at 3-Year Follow-Up of REFLECT Phase III Trial
Regulatory News:
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20230312005028/en/
Graph 1: Evolution of Best-Corrected Visual Acuity

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc

LivaNova erhält 510(k)-Freigabe der US-amerikanischen FDA für Essenz-Herz-Lungen-Maschine für kardiopulmonale Bypassverfahren
LivaNova PLC (Nasdaq: LIVN), ein marktführendes Unternehmen im Bereich medizintechnische Innovationen, gab heute bekannt, dass es die 510(k)-Freigabe von der US-amerikanischen Food and Drug

LivaNova Receives U.S. FDA 510(k) Clearance for Essenz Heart-Lung Machine for Cardiopulmonary Bypass Procedures
LivaNova PLC (Nasdaq: LIVN), a market-leading medical technology and innovation company, today announced it received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Essenz™
EQS-News: Invitation to MorphoSys’ Full Year Results 2022 Conference Call on March 16, 2023
EQS-Adhoc: STRATEC REPORTS PRELIMINARY RESULTS FOR 2022 FINANCIAL YEAR AND ISSUES FINANCIAL GUIDANCE FOR 2023

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc
EQS-News: Drägerwerk AG & Co. KGaA: Dräger expects return to profitable growth in 2023

AEVIS VICTORIA SA – AEVIS VICTORIA achieved consolidated revenues of CHF 1.143bn in 2022, up 27.8% (CHF 895.0m) from prior year.

Form 8.3 - The Vanguard Group, Inc.: Horizon Therapeutics plc







