News

Pfizer and BioNTech Receive U.S. FDA Emergency Use Authorization of COVID-19 Vaccine Booster for Individuals 12 Years of Age and Older
Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced that the U.S. Food and Drug Administration (FDA) has expanded the Emergency Use Authorization (EUA) of a booster dose of the

Pfizer Invites Public to Listen to Webcast of Pfizer Discussion at Healthcare Conference
Pfizer Inc. (NYSE: PFE) invites investors and the general public to listen to a webcast of a discussion with Albert Bourla, Chairman and CEO, at the Goldman Sachs 14th Annual Healthcare CEOs

Pfizer Invites Public to View and Listen to Webcast of February 8 Conference Call with Analysts
Pfizer Inc. (NYSE: PFE) invites investors and the general public to view and listen to a webcast of a conference call with investment analysts at 10 a.m. EST on Tuesday, February 8, 2022. The

Pfizer Receives U.S. FDA Emergency Use Authorization for Novel COVID-19 Oral Antiviral Treatment
Pfizer Inc. (NYSE: PFE) announced today that the U.S. Food and Drug Administration (FDA) has authorized the emergency use of PAXLOVID™ (nirmatrelvir [PF-07321332] tablets and ritonavir tablets) for

Pfizer to Provide the United Kingdom an Additional 2.5 Million Treatment Courses of Investigational Oral Antiviral Candidate to Help Combat COVID-19
Pfizer Inc. (NYSE: PFE) announced an additional agreement with the United Kingdom (UK) government to supply an additional 2.5 million treatment courses of its investigational candidate PAXLOVID™

Pfizer’s VYNDAQEL®/VYNDAMAX® Reduced the Risk of All-Cause Mortality by 41% Among Patients with Transthyretin Amyloid Cardiomyopathy, Five-Year Follow-Up Data Demonstrate
Pfizer Inc. (NYSE: PFE) announced today the publication of a post-hoc, interim analysis showing that treatment with VYNDAQEL® (tafamidis meglumine) / VYNDAMAX® (tafamidis) provided a clinically

Pfizer and BioNTech to Provide European Union More Than 200 Million Additional Doses of COMIRNATY® to Help Meet Continued Need for Vaccine Supply
Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced an agreement has been reached with the European Commission (EC) and its member states to exercise an option to purchase more

EMA Issues Advice for Potential Early Use of Pfizer’s Novel COVID-19 Oral Antiviral Candidate
Pfizer Inc. (NYSE: PFE) announced today that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) issued advice on the use of PAXLOVID™ (nirmatrelvir

U.S. FDA Approves Pfizer’s XELJANZ® (tofacitinib) for the Treatment of Active Ankylosing Spondylitis
Pfizer Inc. (NYSE: PFE) announced today that the U.S. Food and Drug Administration (FDA) has approved the supplemental New Drug Application (sNDA) for XELJANZ® / XELJANZ® XR (tofacitinib) for the

Pfizer Announces Additional Phase 2/3 Study Results Confirming Robust Efficacy of Novel COVID-19 Oral Antiviral Treatment Candidate in Reducing Risk of Hospitalization or Death
Pfizer Inc. (NYSE: PFE) today announced final results from an analysis of all 2,246 adults enrolled in its Phase 2/3 EPIC-HR (Evaluation of Protease Inhibition for COVID-19 in High-Risk Patients)

Pfizer to Acquire Arena Pharmaceuticals
Pfizer Inc. (NYSE: PFE) and Arena Pharmaceuticals, Inc. (Nasdaq: ARNA) today announced that the companies have entered into a definitive agreement under which Pfizer will acquire Arena, a clinical

Pfizer Declares First-Quarter 2022 Dividend
Pfizer Inc. (NYSE: PFE) today announced that its board of directors declared an increase in the quarterly cash dividend on the company’s common stock to $0.40 for the first-quarter 2022 dividend

European Commission Approves Pfizer’s Cibinqo® (abrocitinib) for the Treatment of Adults with Moderate-to-Severe Atopic Dermatitis
Pfizer Inc. (NYSE: PFE) today announced that the European Commission (EC) has approved the 100 mg and 200 mg doses of Cibinqo® (abrocitinib), an oral, once-daily, Janus kinase 1 (JAK1) inhibitor

Pfizer and BioNTech Receive U.S. FDA Emergency Use Authorization of COVID-19 Vaccine Booster for Individuals 16 Years and Older
Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced that the U.S. Food and Drug Administration (FDA) has expanded the Emergency Use Authorization (EUA) of a booster dose of the

Pfizer and BioNTech Provide Update on Omicron Variant
Pfizer Inc. (NYSE: PFE) and BioNTech SE (Nasdaq: BNTX) today announced results from an initial laboratory study demonstrating that serum antibodies induced by the Pfizer-BioNTech COVID-19 Vaccine

Pfizer Invites Public to Register for Webcast of Analyst and Investor Call to Discuss Pfizer-BioNTech COVID-19 Vaccine and Pfizer’s Novel COVID-19 Oral Antiviral Treatment Candidate
Pfizer Inc. (NYSE: PFE) invites investors and the general public to access a live webcast of a presentation and conference call with investment analysts at 8:30 a.m. EST on Friday, December 17

4 Dividend Aristocrats Trading at a Discount
The first quarter of the year was a success for investors. The overall market (NYSE: SPY) surged nearly 10%, fueled by robust economic indicators, optimism, and hype surrounding AI. The

Pfizer’s earnings growth trade at a deep discount, suddenly a buy
The defensive names, characterized by their immunity to the business cycle, in the market could soon see an inflow of investment dollars not seen since the last wave of the business cycle. This

MarketBeat Week in Review – 5/22 - 5/26
Two competing stories are fighting for investors’ attention. The bulls are trying to push the market higher in expectations of a resolution to the debt ceiling standoff. But any excitement is being
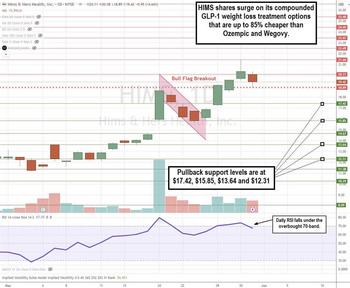
Hims & Hers Health Soars on Generic GLP-1 Rollout Plans
Him & Hers Health Inc. (NYSE: HIMS) provides a direct-to-consumer (DTC) health and wellness platform. The company was best known for prescribing treatments for erectile dysfunction (ED) through

Will the Biotech Sector Shift From Lagger to Leader?
The biotech sector and its popular ETF, the iShares Biotechnology ETF (NASDAQ: IBB), have lagged the overall market during the year, with its shares slightly red. However, in recent weeks, the
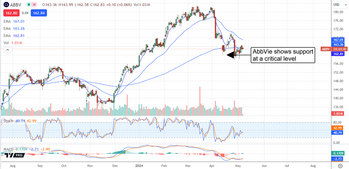
AbbVie Tracking for New Highs in 2024
Shares of AbbVie (NYSE: ABBV) fell hard in the wake of its Q1 earnings report, but the dip is over. The market response to slowing Humira sales is overshadowed by strength in the non-Humira
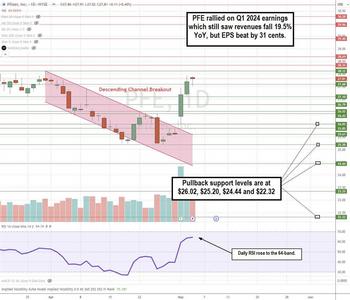
Pfizer Finds a Foothold After a Solid Q1 2024 Earnings Report
Biopharma giant Pfizer Inc. (NYSE: PFE) has given back all its COVID gains and more. Shareholders have sat through a painful two-year selloff from the stock's $61.71 high on December 20, 2021, to

Analyst Sentiment, Revenue Growth Will Lead AbbVie Stock Higher
AbbVie Inc. (NYSE: ABBV) stock is recovering after falling over 4% in the week preceding its earnings report on April 26, 2024. As the market closed on Monday, April 29, ABBV stock posted a gain
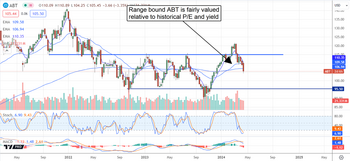
Abbott Laboratories Outlook is Healthy: Buy the Dip
Abbott Laboratories' (NYSE: ABT) share price was corrected at the end of CQ1 2024 on growth, profitability and capital returns concerns. The FQ1 results, however, belie the fear and have the market




